Virtual Urchin Lab
Essential Question: The ocean covers 2/3 of the planet- Is it really possible that humans are changing the chemistry of the ocean?
1) Carbon in the Air: What does the graph of atmospheric Carbon Dioxide tell us?
The graph tells us the CO2 levels measured at the Mauna Loa observatory in Hawaii from 1958-2008.
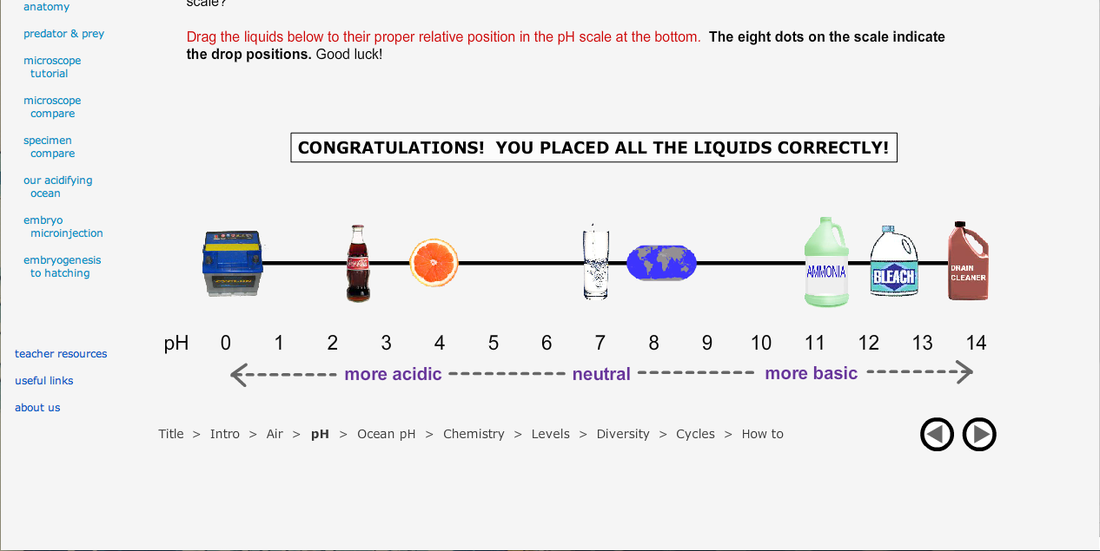
2) pH Scale: Where do some common items fall on the pH scale?
1) Carbon in the Air: What does the graph of atmospheric Carbon Dioxide tell us?
The graph tells us the CO2 levels measured at the Mauna Loa observatory in Hawaii from 1958-2008.
2) pH Scale: Where do some common items fall on the pH scale?
3) Ocean pH: How might it change? A change from 8.2 to 8.1 on the pH scale is a 30% increase in acidity.
4) Carbon in the Water: Explain what happens to carbon in the water.
Carbon in the water becomes carbonic acid, then it bicarbonates, after that it carbonates. Next it becomes calcium carbonate, then turns into a sea urchin or coral.
5) Exploring Carbon Levels and Effects: Look over the interactive and describe what happens at each of the levels:
As the year increases the CO2 increases with the year. The coral calcification gets thinner.
Scenario 1: Optimistic
As the year increases the CO2 increases with the year. The coral calcification gets thinner.
Scenario 2: Middle Ground
The CO2 increase more and the pH levels decrease a lot. The coral calcification makes it thinner too.
Scenario 3: Pessimistic
The CO2 increases alot more and the pH levels decreases alot. The coral calcification is alot thinner.
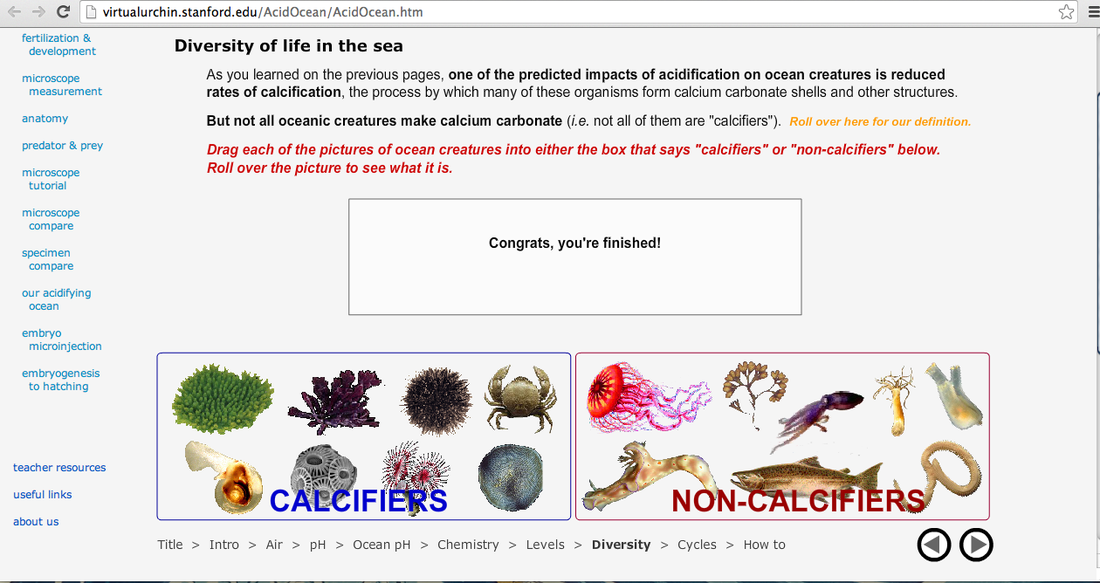
6) Diversity of Life in the Sea: Sort each of the organisms into Calcifiers or Non-Calcifiers.
4) Carbon in the Water: Explain what happens to carbon in the water.
Carbon in the water becomes carbonic acid, then it bicarbonates, after that it carbonates. Next it becomes calcium carbonate, then turns into a sea urchin or coral.
5) Exploring Carbon Levels and Effects: Look over the interactive and describe what happens at each of the levels:
As the year increases the CO2 increases with the year. The coral calcification gets thinner.
Scenario 1: Optimistic
As the year increases the CO2 increases with the year. The coral calcification gets thinner.
Scenario 2: Middle Ground
The CO2 increase more and the pH levels decrease a lot. The coral calcification makes it thinner too.
Scenario 3: Pessimistic
The CO2 increases alot more and the pH levels decreases alot. The coral calcification is alot thinner.
6) Diversity of Life in the Sea: Sort each of the organisms into Calcifiers or Non-Calcifiers.
7) Echinoderms Life Cycles and Skeletons: Describe the life cycle of Echinoderms and explain why they are so sensitive to changes in pH.
First the 2 cell embryo, is off free of parental supervision on its own. Second, it turns into a young sea urchin larvae and find
8) How to study Ocean Acidification in the Lab- How may ocean acidification impact sea urchins in the larval stage? Can you think of any other possible acidification impacts on marine organisms other than calcification?
First the 2 cell embryo, is off free of parental supervision on its own. Second, it turns into a young sea urchin larvae and find
8) How to study Ocean Acidification in the Lab- How may ocean acidification impact sea urchins in the larval stage? Can you think of any other possible acidification impacts on marine organisms other than calcification?
1: Complete the pre-lab certification.
What I see is a circle that kind of looks like an eye. There seems to be something little particles towards the right side of the circle.
2: Look at the first slide under the microscope - Describe what you see.
We are using these two pH values for this experiment because pH 8.1 reflects the pH level today, and pH 7.7 reflects the pH level at the end of the century.
3: Fill two flasks with sea water and label them with "pH 7.7" and "pH 8.1". Explain why we are using these two pH values for this experiment.
4: What happens to the pH of the first sample when Carbon Dioxide is added? Explain.
The pH of the first sample becomes lower.
5: Complete the rest of the procedures to prepare slides for study
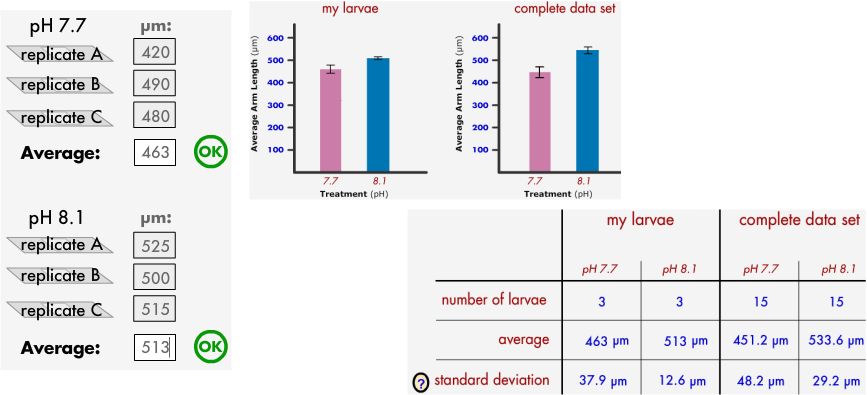
6: After making measurements- Find the AVERAGE of each data set
463 for pH 7.7 & 513 for pH 8.1
Take a screen shot of the data data/graphs and paste below:
What I see is a circle that kind of looks like an eye. There seems to be something little particles towards the right side of the circle.
2: Look at the first slide under the microscope - Describe what you see.
We are using these two pH values for this experiment because pH 8.1 reflects the pH level today, and pH 7.7 reflects the pH level at the end of the century.
3: Fill two flasks with sea water and label them with "pH 7.7" and "pH 8.1". Explain why we are using these two pH values for this experiment.
4: What happens to the pH of the first sample when Carbon Dioxide is added? Explain.
The pH of the first sample becomes lower.
5: Complete the rest of the procedures to prepare slides for study
6: After making measurements- Find the AVERAGE of each data set
463 for pH 7.7 & 513 for pH 8.1
Take a screen shot of the data data/graphs and paste below:
Discuss what your data showed: So What?
Sea urchin larvae in water that is more acidic grows less.
Conclusion: What would these changes do to the adult Sea Urchin population?
They might be small, sick or die early.
How would that effect other organisms that depend on these organisms as a source of food? (Sea Otters)?
They will have less to eat.
What did you learn about Ocean Acidification? Discuss.
I learned that it doesn't take a lot of change in pH to affect sea life, and that pH change in the ocean, by hurting small organisms, can mess up the entire ecosystem.
Sea urchin larvae in water that is more acidic grows less.
Conclusion: What would these changes do to the adult Sea Urchin population?
They might be small, sick or die early.
How would that effect other organisms that depend on these organisms as a source of food? (Sea Otters)?
They will have less to eat.
What did you learn about Ocean Acidification? Discuss.
I learned that it doesn't take a lot of change in pH to affect sea life, and that pH change in the ocean, by hurting small organisms, can mess up the entire ecosystem.