Marine Science - Water Notes
What is water?
- Two parts ( atoms ) hydrogen, one part oxygen
- An atom is the smallest unit of matter that retains its properties.
- Atoms consist of protons, electrons , and neutrons
-Protons have a positive charge
-Electrons have a negative charge
-Neutrons are all neutrally charged ( no charge )
Atoms are usually neutrally charged because the number of protons and electrons are even
Unevenly charged atoms are called ions.
When two hydrogen atoms and one oxygen atom combine to form a water molecule, each hydrogen atom shares its single electron with the oxygen, and the oxygen shares one of its electrons with the hydrogen.
- Two parts ( atoms ) hydrogen, one part oxygen
- An atom is the smallest unit of matter that retains its properties.
- Atoms consist of protons, electrons , and neutrons
-Protons have a positive charge
-Electrons have a negative charge
-Neutrons are all neutrally charged ( no charge )
Atoms are usually neutrally charged because the number of protons and electrons are even
Unevenly charged atoms are called ions.
When two hydrogen atoms and one oxygen atom combine to form a water molecule, each hydrogen atom shares its single electron with the oxygen, and the oxygen shares one of its electrons with the hydrogen.
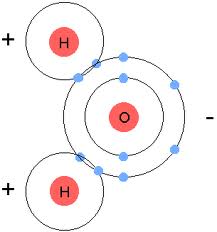
Water is a Polar Molecule
Shared electrons spend more time around the oxygen atom than the hydrogen atom. The hydrogen atom carries a slightly positive charge while the oxygen atom carries a slightly negative charge. In other words, the oxygen atom is negative and the hydrogen atom is positive. Because of that, the oxygen atom and the hydrogen atoms are attracted to each other.
Shared electrons spend more time around the oxygen atom than the hydrogen atom. The hydrogen atom carries a slightly positive charge while the oxygen atom carries a slightly negative charge. In other words, the oxygen atom is negative and the hydrogen atom is positive. Because of that, the oxygen atom and the hydrogen atoms are attracted to each other.
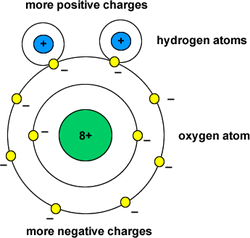
Hydrogen bonding is a weak attraction between the weakly positive hydrogen atoms and the weakly negative oxygen atoms of a different molecule. Hydrogen bonding is responsible for the unique properties of water.
Water is a Universal Solvent
Polarity of water gives it the ability to dissolve most compounds. The positive hydrogen side of water is attracted to the negative parts of the compound it is dissolving. Negative oxygen is attracted to the positive parts. This allows water to dissolve and break apart ionically charged substances. Cohesion is when water is attracted to other water molecules. This helps trees pull water up into their branches. Water is able to pull itself into a shape with the smallest amount of surface area. Surface tension is the attraction of water molecules to themselves helps to create a "film" on the surface of bodies of water. Small critters can "walk" across the water. Adhesion is when water is attracted to other materials.
Polarity of water gives it the ability to dissolve most compounds. The positive hydrogen side of water is attracted to the negative parts of the compound it is dissolving. Negative oxygen is attracted to the positive parts. This allows water to dissolve and break apart ionically charged substances. Cohesion is when water is attracted to other water molecules. This helps trees pull water up into their branches. Water is able to pull itself into a shape with the smallest amount of surface area. Surface tension is the attraction of water molecules to themselves helps to create a "film" on the surface of bodies of water. Small critters can "walk" across the water. Adhesion is when water is attracted to other materials.
States of Matter
Solid (ice), Liquid (water), and Gas (water vapor). Earth is the only planet in the solar system where water is found in liquid form. Temperature of a substance is a measure of average kinetic energy of the atoms and molecules measured in degrees. Heat is a measure of the total kinetic energy of the atoms and molecules measured in Calories. One calorie is equal to the amount of heat needed to raise the temperature of one gram of water by one degree Celsius. When you eat one hundred calories, you body needs energy to burn them off. When your body can't burn calories off, they store them off as fat.
Solid (ice), Liquid (water), and Gas (water vapor). Earth is the only planet in the solar system where water is found in liquid form. Temperature of a substance is a measure of average kinetic energy of the atoms and molecules measured in degrees. Heat is a measure of the total kinetic energy of the atoms and molecules measured in Calories. One calorie is equal to the amount of heat needed to raise the temperature of one gram of water by one degree Celsius. When you eat one hundred calories, you body needs energy to burn them off. When your body can't burn calories off, they store them off as fat.

The pool is big, so it has more heat.

The boiling pot is small, but it has higher temperature.
Effect of Heat on Water
High heat of vaporization - Hydrogen bonds must be broken so water can absorb a lot of heat without charging phase. It evaporates slowly and that causes the cooling effect of sweating.
High heat of vaporization - Hydrogen bonds must be broken so water can absorb a lot of heat without charging phase. It evaporates slowly and that causes the cooling effect of sweating.
Water has a specific heat - This means that water can absorb a large amount of heat before it begins to get hot. The specific heat of water also helps regulate the rate at which air changes temperature, which is why the temperature change of seasons is so gradual but not sudden, especially near large bodies of water.
What is Density?
Density is a physical property of a substance that describes the degree of compactness of a substance- in other words, how closely packed the atoms are. The more closely packed the atoms are in a substance, the more dense it is.
Density= Mass/ Volume
What is Density?
Density is a physical property of a substance that describes the degree of compactness of a substance- in other words, how closely packed the atoms are. The more closely packed the atoms are in a substance, the more dense it is.
Density= Mass/ Volume
Investigating Water Lab
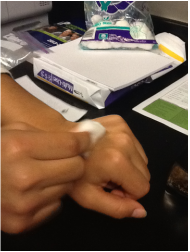
Evaporative Cooling Station # 1 Water and Alcohol
My group thought that the alcohol was cooler than the water. It is beneficial to sweat water than alcohol because water cools you down faster and stays longer than alcohol.
My group thought that the alcohol was cooler than the water. It is beneficial to sweat water than alcohol because water cools you down faster and stays longer than alcohol.

Water Tension/ Cohesion Station #2 Water on a Penny
It took 24 drops to fit on the penny. The penny was able to hold that many drops of water because of cohesion.
It took 24 drops to fit on the penny. The penny was able to hold that many drops of water because of cohesion.

Surface Tension Station #3 Water on Wax Paper
When we put the drop of water onto the wax paper the water turned into round beads. The water turned into beads because of surface tension.
When we put the drop of water onto the wax paper the water turned into round beads. The water turned into beads because of surface tension.

Surface Tension Station #4 Paper Clips on the Surface of a Cup of Water
In this station we had to try to get paperclips to float on top of the water surface before it sunk but unfortunately we couldn't get a paperclip to float.
In this station we had to try to get paperclips to float on top of the water surface before it sunk but unfortunately we couldn't get a paperclip to float.

Adhesion Station #5 Graduated Cylinder
The water is not a straight line across in the graduated cylinder because of adhesion. The water property of adhesion is important in living systems because it helps them survive and they really depend on it.
The water is not a straight line across in the graduated cylinder because of adhesion. The water property of adhesion is important in living systems because it helps them survive and they really depend on it.

Capillary Action Station #6 Rolled up Paper Towel
The water is transferring from one cup to another because of capillary action. Capillary action would be important to plants and trees because they need water to survive and helps pull the water up the plants to live.
The water is transferring from one cup to another because of capillary action. Capillary action would be important to plants and trees because they need water to survive and helps pull the water up the plants to live.